
An international research collaboration led by scientists at City University of Hong Kong (CityU) has explained a long-standing thermodynamic inconsistency in the formation of a class of metallic glass that may lead to the development of new, better metallic alloys.
Metallic glass is considered to be an advanced material because of its exceptional physical properties, such as superior strength, hardness, wear, resistance to corrosion and formability. It is used in a range of applications, such as medical devices, transformers and sports equipment.

Professor Xun-Li Wang, Chair Professor of Physics and Head of the Department of Physics and Materials Science of CityU, who headed the project, said the discovery of a hidden amorphous phase in palladium-nickel-phosphorus metallic glass is an important observation in glass physics.
The research has just been published in the prestigious journal Nature Communications.
“We can now explore how to produce or induce this amorphous phase in metallic glass, so that we can tune the properties of the material in larger sizes for better applications,” said Professor Wang.
A long-standing issue in metallic glass research had been to resolve the packing of atoms in the material because the structure determines properties.
Unlike most metals, where the atoms are packed into regular arrays, metallic glasses are composed of atoms in a disordered arrangement; it is this so-called amorphous structure that imparts valuable properties to these materials.
In a world-first experiment, performed at the Australian Centre for Neutron Scattering, a crucial hidden amorphous phase was revealed.
|
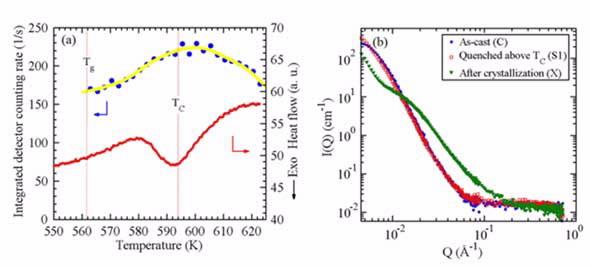
The results of simultaneous DSC-SANS measurements for the Pd41.25Ni41.25P17.5 alloy. (a) The integrated detector counting rates as a function of temperature. The simultaneous DSC scan is superimposed, which shows a TC ~ 594 K at a heating rate of 2.5 K/min. The co-existence of two phases can be clearly seen in the vicinity of Tc. (b) Full-Q range SANS data for three samples: as-cast condition (C), quenched from 623 K after the transformation had ended (S1), and quenched after crystallization at 673 K (X). The nearly complete overlap of the SANS profiles between the as-cast and S1 samples demonstrates the reentrant behavior.
“We were measuring the small-angle neutron scattering (SANS) while heating the glass to assess changes in its structure while, simultaneously, measuring changes in the amount of heat the material was absorbing using a method called differential scanning calorimetry,” said QUOKKA instrument scientist, Dr Elliot Gilbert, a co-author on the paper.
“You simply can’t perform this measurement anywhere else in the world. The special device used in the experiments was developed here at ANSTO. We’re the only facility in the world in which SANS and differential scanning calorimetry can be measured at the same time.
Typically the way one might do these experiments is to take the material and study it with a range of different techniques, but unfortunately, not at the same time. When trying to relate the structural changes occurring during heating, you simply can’t be sure that the data you collect from one measurement can be related to another if they are collected using different temperature probes or at different times, perhaps months apart.
We were able to directly correlate changes in the structure of the material with the energy required for that structure to change,” said Gilbert.
Synchrotron X-ray measurements at Argonne National Laboratory provided information on the atomic length scale that showed a re-arrangement of atomic clusters with temperature. This was complemented by studies at Hokkaido University in Japan where high resolution microscopy images and electron diffraction patterns were acquired.
“By bringing together researchers from around the world, this discovery opens a way to manipulate the processing conditions of these materials, such as heat treatment, to generate advantageous behavior,” said Gilbert.
