ANSTO has contributed to a recent publication in Nature Communications Biology that represents a landmark achievement in structural biology: an understanding of protein regulation mechanisms in the bacterium, Mycobacterium tuberculosis (Mtb), the causative agent of tuberculosis (TB) a major global health threat.
A research team led by the University of Melbourne combined several advanced techniques at the Australian Synchrotron and the NCRIS-funded National Deuteration Facility (PDF, 3.13 MB) to reveal the hidden “allosteric” mechanism that activates a key enzyme, ICL2.
The study opens a target pathway to treat drug-resistant TB with modulators that can interfere with the enzyme’s ‘on switch.’ Traditional drugs often targeted the enzyme’s active site, which is difficult to block effectively.
However, ICL2 is unique to mycobacteria and is essential for the survival of the TB bacterium during infection, especially when it is starved of sugar and forced to live on fats.
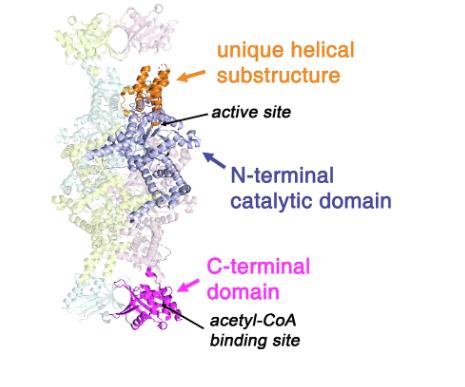
The Macromolecular Crystallography (MX2) beamline was instrumental in determining the crystal structure of an inactive form of the ICL2 enzyme at a high resolution. This allowed researchers to see the exact positions of atoms and confirm the formation of specific chemical bonds (disulfide bridges) used to lock the enzyme in a particular shape; called the “fixed” version.
The Small-angle X-ray scattering (SAXS/WAXS) beamline was used to understand the enzyme’s behaviour in its natural, fluid state. It revealed that without its activator, the enzyme is highly flexible and has multiple orientations.
“This inherent flexibility meant the natural inactive state of the enzyme could not be crystallised. It further highlights how biological SEC-SAXS in solution can support the understanding of these flexible systems and explains why the enzyme is less active in that state”, said Beamline Group Manager Dr Ashish Sethi, a co-author on the paper.
The combined use of MX2 and biological SAXS techniques allowed the team to see not just what the enzyme looks like, but exactly how it moves and how it is controlled.
To overcome technical hurdles in nuclear magnetic resonance (NMR) imaging, a team at the National Deuteration Facility (NDF) produced a partially deuterated form of the protein.
This precision labelling allowed investigators to see dynamic changes that are otherwise invisible, allowing the team to confirm that the enzyme forms two identical molecules, a dimer, when it is activated.
To enable nuclear magnetic resonance (NMR) spectroscopy measurements capable of revealing dynamic changes that would otherwise remain invisible, the National Deuteration Facility (NDF) produced both multiple stable isotope labelled (2H, 13C, 15N: partially deuterated) and double-labelled (2H,15N: perdeuterated) forms of the protein, utilising recombinant bacterial protein expression techniques.”
Deuteration together with the 13C and 15N labelling enabled NMR measurements to provide the information needed to determine the connectivity and environment of the atoms” said NDF Operations Manager, Ms Karyn Wilde, a co-author on the paper.
Other collaborating organisations included The University of Auckland and Monash University.
According to the WHO, 1.5 million people die from TB each year. Approximately 5.8 per 100,000 people developed the disease from 2015-2018, according to estimates in the Journal of Communicable Diseases Intelligence. Australia’s overseas-born population continue to represent the majority of TB notifications (between 86% to 89% across the four reporting years) and the Aboriginal and Torres Strait Islander population continues to record TB rates around four to five times higher than the Australian-born non-Indigenous population.









