
What are radioisotopes?
What is an isotope?
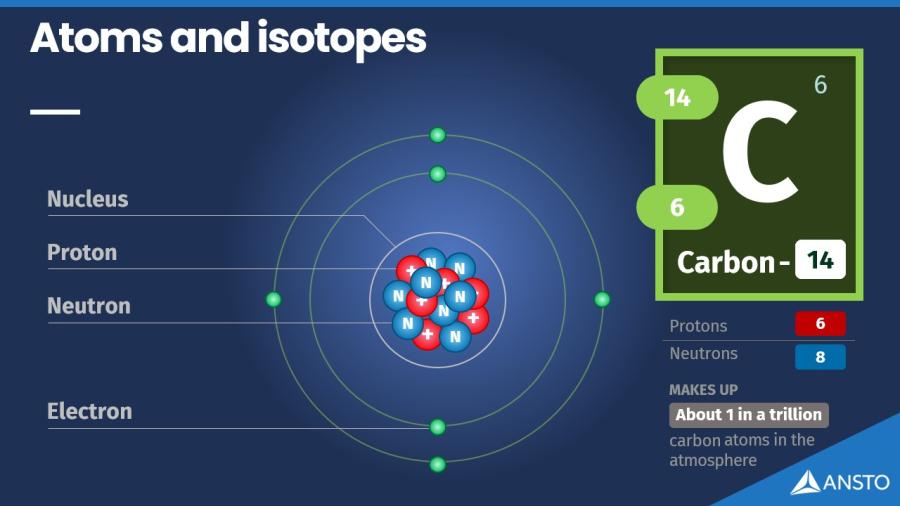
Isotopes are atoms of the same element with different numbers of neutrons in their nuclei.
Atoms are the tiny building blocks that make up everything around us. Each atom has three main parts:
- Protons, which have a positive charge
- Neutrons, which have no charge
- Electrons, which have a negative charge and move around the outside of the atom
The number of protons in an atom tells us what element it is. For example, all carbon atoms have six protons.
Atoms of the same element can have different numbers of neutrons, and this is what creates isotopes. They behave the same chemically, but they have slightly different masses.
For example:
- Carbon-12 has 6 protons and 6 neutrons
- Carbon-13 has 6 protons and 7 neutrons
- Carbon-14 has 6 protons and 8 neutrons
Carbon-12 and carbon-13 are stable and make up almost all the carbon on Earth. Carbon-14 has an extra neutron, which makes it unstable. Because of this, it slowly breaks down over time, releasing energy. When an isotope is unstable like this, we call it a radioisotope.

Learning resources
Here are some resources to help you learn more.
What is a radioisotope?
Radioisotopes are simply unstable isotopes.
Some isotopes are stable, meaning their nuclei stay together forever. Others have a nucleus that is a bit unbalanced. These isotopes naturally try to become more stable by releasing small amounts of energy or tiny particles. When an isotope does this, we call it a radioisotope.
This process of becoming more stable is called radioactive decay. Different radioisotopes release different kinds of radiation as they decay.
Types of radiation a radioisotope can emit
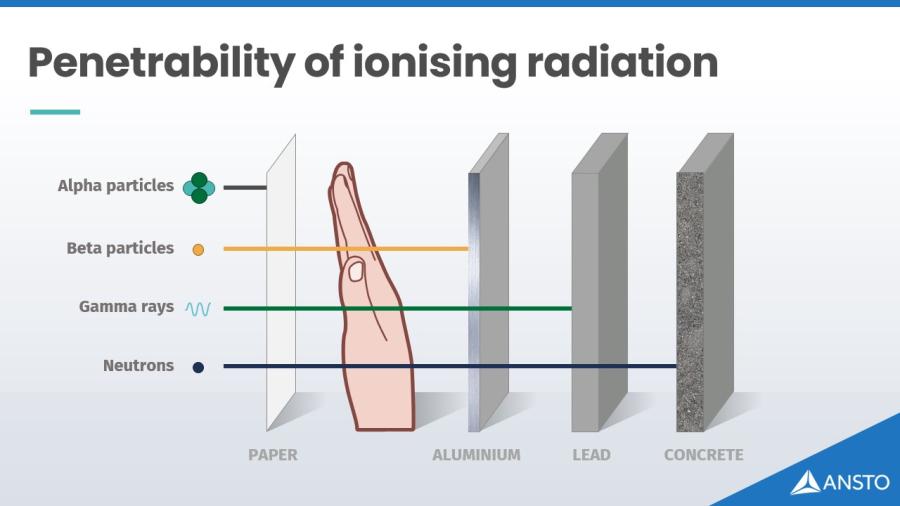
Radioisotopes may give off:
- Alpha particles – heavy, slow particles that can be stopped by paper
- Beta particles – fast, light electrons that travel further but can be blocked by thin metal
- Gamma rays – high energy waves, similar to Xrays, which can travel through the body and need thicker shielding energy waves, rays, which can travel through the body and need thicker shielding
These forms of radiation happen naturally whenever an unstable nucleus rearranges itself.
Why radioisotopes decay
A nucleus can be unstable for a few reasons:
- It may have too many neutrons
- It may have not enough neutrons
- It may be too large to hold itself together
- It may contain too much excess energy
When it releases radiation, the nucleus changes into a different, more stable isotope.
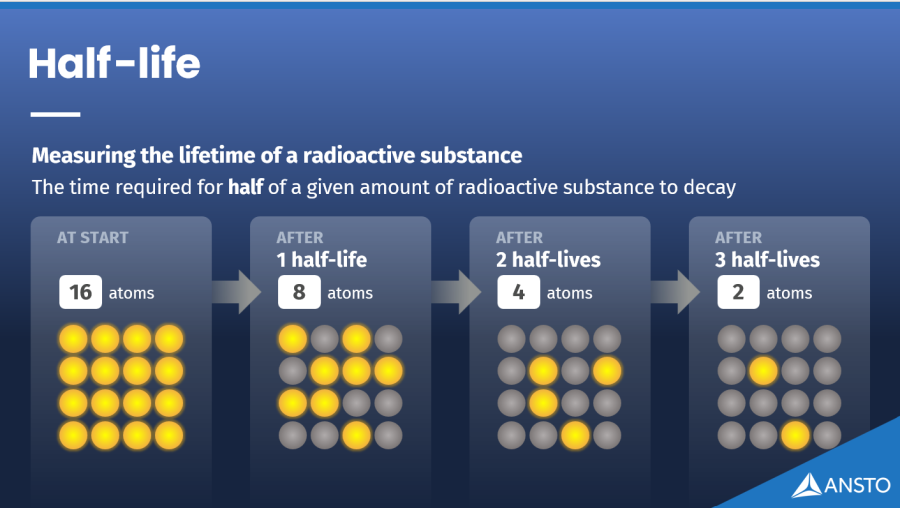
How fast do radioisotopes decay?
Each radioisotope decays at its own natural rate. Scientists describe this using a term called half-life, which is the time it takes for half of a sample to decay. Some radioisotopes decay in seconds, while others take thousands or millions of years.
Radioisotopes are useful
Because radioisotopes give off radiation in predictable ways, they can be used for many purposes, from medical imaging to tracing water through the environment.
You are naturally radioactive
Natural radioisotopes are present in the human body
Cosmic radiation in flight
Radiation exposure increases at high altitude
Radioisotopes used for medical imaging
Technetium-99m is widely used to image organs
Where do we find isotopes and radioisotopes?
Isotopes are everywhere. Every element on the Periodic Table has more than one isotope, and many of these occur naturally in our environment, our food and even our own bodies.
In the Earth
Rocks, minerals and soil contain natural isotopes that have been present since the planet formed. Some of these are stable, while others are longlived radioisotopes such as:
- Uranium and thorium, found in many types of rock
- Potassium-40, found in soil and in some foods
- Natural decay products like radon gas, which can collect indoors in small amounts
These all contribute to naturally occurring background radiation (the radiation exposure we get from radioisotopes found in our environment) which we are exposed to every day.
In the atmosphere and from space
The atmosphere contains isotopes created naturally, including:
- Carbon-14, produced when cosmic rays interact with atoms high in the atmosphere
- Cosmic radiation, which constantly reaches Earth from the sun and space
Our atmosphere acts as a protective shield, so the amount of cosmic radiation we receive at ground level is low.
However, when you fly in an aeroplane, you are above much of this protective atmosphere, so you receive a slightly higher dose of cosmic radiation during the flight. It is still a small amount —, roughly the same as several days of natural background radiation —, but it’s a helpful example of how radiation levels vary depending on altitude.
In our food and our bodies
Many of the elements we eat or drink have natural radioisotopes. For example:
- Bananas, nuts and many vegetables contain small amounts of the natural radioisotope potassium-40
- Our bodies naturally contain radioisotopes of carbon, such as carbon-14, along with stable isotopes of carbon, nitrogen and oxygen
These natural radioisotopes are present in very small amounts and are a normal part of healthy biological processes.
In everyday objects
Some materials and products also contain small amounts of naturally occurring radioactive elements, such as:
- Older luminous watches and clocks
- Certain types of pottery or glassware
- Fertilisers that contain potassium
These levels are extremely low and sit comfortably within normal background exposure.

Image: Uranium glassware infused with uranium oxide created between the 1830s and 1930s.
How radioactive am I?
Find out sources of background radiation and estimate your yearly radiation exposure with our fun quiz
How do radioisotopes get used?
Radioisotopes are valuable tools in medicine, environmental science and industry.
Because they emit small, measurable amounts of radiation, they can act as tracers or targeted sources of energy that make it possible to measure or image things that cannot normally be seen.
ANSTO manufactures radioisotopes for use in healthcare, research and industry.

Image: Scientist developing radioisotopes at ANSTO.
Nuclear medicine (manufactured radioisotopes)
ANSTO manufactures radioisotopes used in nuclear medicine for both diagnosis and treatment.
Diagnosis (medical imaging)
Some manufactured radioisotopes can act as tracers inside the body. The small amounts of radiation they emit can be detected by specialised cameras, allowing doctors to see how organs and tissues are functioning.
A widely used example is technetium-99m (Tc99m), which is used for imaging organs and soft tissues such as the lungs, bone, brain, liver and kidneys.
Treatment (targeted radiation therapy)
Some manufactured radioisotopes are used to treat disease by delivering radiation to specific tissues. A common example is iodine-131 (I131), which is used to treat certain thyroid conditions because iodine naturally concentrates in thyroid tissue.
Tracing radioisotopes in the environment
Radioisotopes can be used as tracers to understand how natural systems behave and change over time. Depending on the question, studies may use naturally occurring radioisotopes, manufactured radioisotopes, or a combination of both.
ANSTO has specialised, radiation licensed facilities that support radioisotope tracing studies in controlled conditions, including work with freshwater and marine organisms and plant studies. licensed facilities that support radioisotope tracing studies in controlled conditions, including work with freshwater and marine organisms and plant studies.
Groundwater
Radioisotopes can help trace how groundwater flows underground, how quickly it is replenished and where different water sources mix. Groundwater age dating can use naturally occurring radioisotopes such as tritium (hydrogen-3), radiocarbon (carbon-14) and chlorine-36 to understand how long water has been in an aquifer and how quickly it recharges.
Environmental change over time
Radioisotopes are also used more broadly in environmental science to examine long term processes such as sediment movement, erosion and changes recorded in natural materials.
Industry and materials science
Radioisotopes and radiation based techniques are widely used across industry to test, measure and improve materials and products. Many of these applications take place within industrial facilities rather than at ANSTO.based techniques are widely used across industry to test, measure and improve materials and products. Many of these applications take place within industrial facilities rather than at ANSTO.
Common applications include:
Nondestructive testing
Radiation is used to inspect welds, components and structures without cutting them open.
Thickness and density measurement
Radioisotopes help manufacturers measure thickness, density or uniformity in materials such as metals, plastics or paper.
Sterilisation
Radiation can sterilise medical products, research materials and specialised items.
GATRI (Gamma Technology Research Irradiator) is located at the Lucas Heights campus and provides controlled gamma irradiation services for research and industry applications such as product testing, quality assurance, materials modification and scientific studies.
Learning resources
Here are some resources to help you learn more.
Day in the life of Australian Nuclear Medicine
Go behind the scenes as nuclear medicine makes its way from the OPAL reactor to medical centres around Australia.
How does ANSTO make radioisotopes?
Radioisotopes used in medicine, research and industry are produced at ANSTO’s OPAL multipurpose research reactor. OPAL produces neutrons that can be used to transform stable atoms into useful radioisotopes.
Using neutrons to create radioisotopes
Inside OPAL, selected target materials are bombarded with neutrons. When a nucleus absorbs a neutron, it often becomes a different isotope of the same element, which may be a radioisotope suitable for imaging, therapy or environmental tracing.
Careful processing and packaging
After irradiation, radioisotopes are transferred to specialised laboratories for processing. These laboratories use shielded facilities and remote handling tools to safely prepare radioisotopes in forms suitable for medical, environmental or industrial use, and to package them for transport.handling tools to safely prepare radioisotopes in forms suitable for medical, environmental or industrial use, and to package them for transport.
Delivery across Australia and internationally
Many radioisotopes have short half-lives, so coordinated production and delivery schedules ensure that hospitals, researchers and industry receive them while they are still active.
The importance of the OPAL reactor
The OPAL reactor provides a reliable source of neutrons for radioisotope production, scientific research and materials investigation. Its design allows Australia to maintain a stable, local supply of essential radioisotopes, reducing reliance on overseas production and supporting critical health and research services.
Radioisotopes created by OPAL reactor
By irradiating targets in the world-class OPAL research reactor, a diverse set of radioisotopes, primarily for nuclear medicine, are available for a variety of applications and for research.
Learning resources
Here are some resources to help you learn more.
FAQs
1. What is the difference between an isotope and a radioisotope?
On the periodic table there are 118 elements, and each element is defined by the number of protons in its nucleus. Atoms of the same element always have the same number of protons, but they can have different numbers of neutrons. These variations are called isotopes.
Most isotopes are stable, meaning their nuclei stay the same over time.
Some isotopes have an unstable nucleus. These isotopes naturally change to become more stable, and in the process, they release small amounts of energy or particles. These unstable isotopes are known as radioisotopes.
2. Where are radioisotopes found?
Radioisotopes occur naturally in many parts of the environment and can also be created for specific uses. Natural radioisotopes have been present on Earth since it formed and continue to be produced through natural processes.
In the environment
Radioisotopes are found in rocks and soil, including long-lived radioisotopes such as uranium, thorium and potassium-40. Small amounts are also present in the atmosphere, where radioisotopes like carbon-14 are created when cosmic rays interact with atoms. Natural radioisotopes can also be found in food, including trace amounts of potassium-40 in bananas, nuts and many vegetables.lived radioisotopes such as uranium, thorium and potassium-40. Small amounts are also present in the atmosphere, where radioisotopes like carbon-14 are created when cosmic rays interact with atoms. Natural radioisotopes can also be found in food, including trace amounts of potassium-40 in bananas, nuts and many vegetables.
From space
Cosmic radiation from the sun and stars interacts with the upper atmosphere, creating radioisotopes such as carbon-14. Levels at ground level are low because the atmosphere provides protection, but exposure increases slightly at high altitudes, such as during air travel.
In the body:
Because food and water contain naturally occurring radioisotopes, small amounts are also found inside the human body. These levels are normal and part of everyday life.
Made radioisotopes
Radioisotopes can also be produced intentionally for use in medicine, research and industry. These manufactured radioisotopes are created in places such as ANSTO’s OPAL reactor and are designed for specific applications, such as medical imaging.
3. How does ANSTO make radioisotopes?
Radioisotopes are made at the Lucas Heights campus using the OPAL research reactor. OPAL produces neutrons that bombard selected materials, turning some of their atoms into useful radioisotopes.
After irradiation, the materials are taken to specialised laboratories where the radioisotopes are separated, purified and prepared for use in medicine, research or industry. Because many radioisotopes decay quickly, production and delivery are timed so they reach hospitals and researchers while still active.
The OPAL reactor provides a reliable source of neutrons, allowing Australia to maintain a secure supply of essential radioisotopes.
4. Are radioisotopes safe?
Radioisotopes can be used safely when proper controls are in place. Their use in medicine, research and industry is closely regulated in Australia to ensure people and the environment are protected. Safety measures include strict handling procedures, specialised shielding, controlled work areas and careful monitoring.
Most radioisotopes used in health care have short half-lives. This means they lose their activity quickly, which helps limit exposure. When used in medical imaging or treatment, the amounts involved are small and are chosen so that the health benefits outweigh any risk.
Radioisotopes that remain active for longer periods are stored, transported and disposed of under strict guidelines set by national authorities. These controls help ensure that radioisotopes can be used effectively while keeping exposure as low as reasonably achievable.
5. What is half-life and why does it matter?
Half-life is the time it takes for half of any given amount of a radioisotope to decay into another form. Every radioisotope has its own half-life, which can range from fractions of a second to millions of years.
Half-life matters because it shows how long a radioisotope will remain active. Short lived radioisotopes are useful in medicine because they deliver radiation for a short time and then decay quickly, reducing long term exposure. Radioisotopes with longer half-lives are used in research and industry under strict controls to ensure safe handling and storage.lived radioisotopes are useful in medicine because they deliver radiation for a short time and then decay quickly, reducing longterm exposure. Radioisotopes with longer half-lives are used in research and industry under strict controls to ensure safe handling and storage.
Understanding half-life helps determine how radioisotopes are produced, transported, used and safely managed.
6. Is radiation always harmful?
Radiation is a natural part of the world and is present everywhere, including in soil, food, the atmosphere and the human body. Most everyday exposure is very low.
Whether radiation is harmful depends on the dose and how it is received. At higher doses, radiation can damage cells and tissues and may cause health effects. These are sometimes called tissue reactions, and they become more likely and more severe as dose increases.
At lower doses, the main concern is a different type of risk, where effects such as cancer can occur by chance. The likelihood of this type of effect increases with dose, even though the risk at everyday background levels is very small.
This is why radiation is carefully controlled in settings such as medicine, research and industry. Medical scans and treatments use planned, measured doses where the health benefits outweigh any risk, and safety systems are designed to keep exposure as low as reasonably achievable.
7. Do radioisotopes stay radioactive for a long time?
The length of time a radioisotope remains radioactive depends on its half-life, which is the time it takes for half of any given amount to decay into another form. Some radioisotopes decay very quickly, while others remain active for much longer.
A practical guide is that after about ten half-lives, more than 99.9 percent of the original radioisotope has decayed. This means:
- Radioisotopes with short half-lives are active for a short time and are used in situations where they need to decay away quickly, such as medical imaging.
- Radioisotopes with longer half-lives remain active and useful for longer periods and are applied in research and industry under strict safety controls.
Understanding how long a radioisotope stays active helps determine how it is used, handled and safely managed.