Micro-Particle Induced X-ray Emission (µPIXE) s based on the same principles as PIXE.
Basic physical principles
As a charged particle moves through a material, it loses energy primarily by exciting electrons in the atoms that it passes by. Electrons in the inner shells of the atom (predominantly the K and L shells) are given enough energy to cause them to be ejected, resulting in an unstable electron atomic configuration.
Electrons from higher shells in the atom then 'drop down' to fill these vacancies, and in so doing, give off excess energy in the form of X-rays. The energies of these X-rays are characteristic of the element and therefore can be used to identify elemental composition. On the other hand, by measuring intensities of characteristic X-ray lines one can determine concentrations of almost all elements in the sample down to approximately 1 ppm (part-per-million).
µPIXE technique
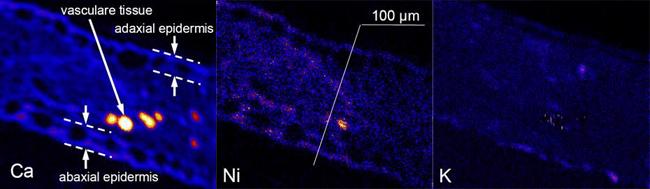
The only difference in µPIXE is that the ion beam is focussed to a small spot size which is then raster-scanned across the sample's surface. In this way, a PIXE spectrum is collected for each raster point. From these spectra, elemental maps can be constructed, that show the variation of an element's concentrations across the sample surface.

Figure 1 Elemental maps for calcium, nickel and potassium from a section of the leaf of a hyperaccumulator plant
Applications
The µPIXE technique can be applied to a wide variety of sample materials. At ANSTO, µPIXE is often used for quantitative analysis in geology, biology, and environmental science.
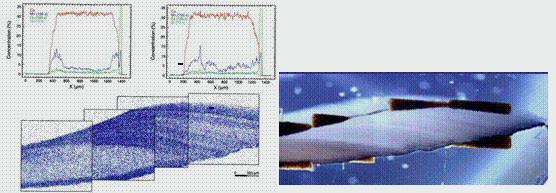
Figure 2 (left) uPIXE analysis with the corresponding micrograph of a bivalve mussel shell cross-section; (right) micrograph size 3x10mm
