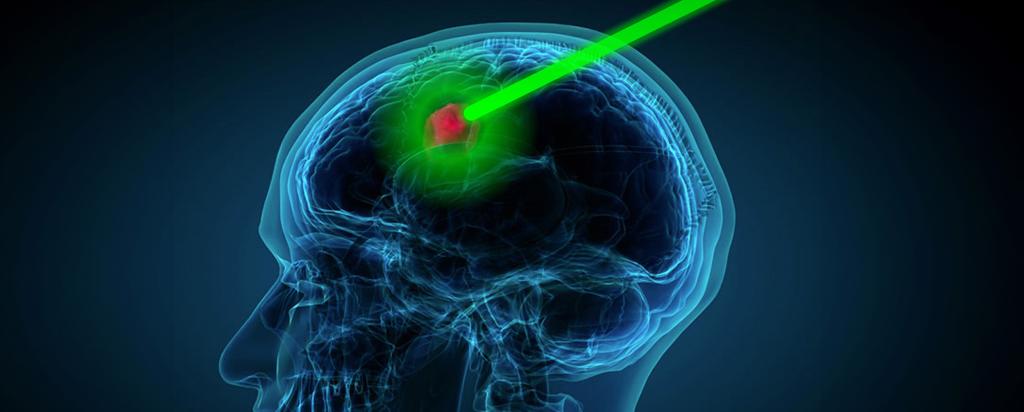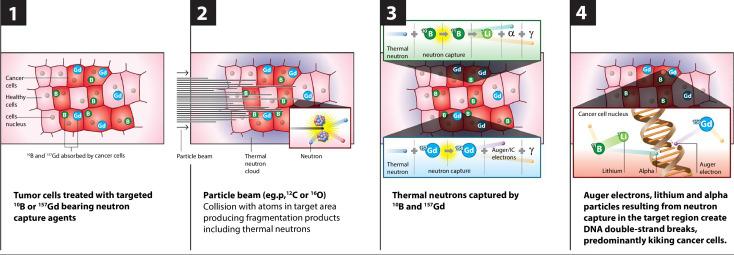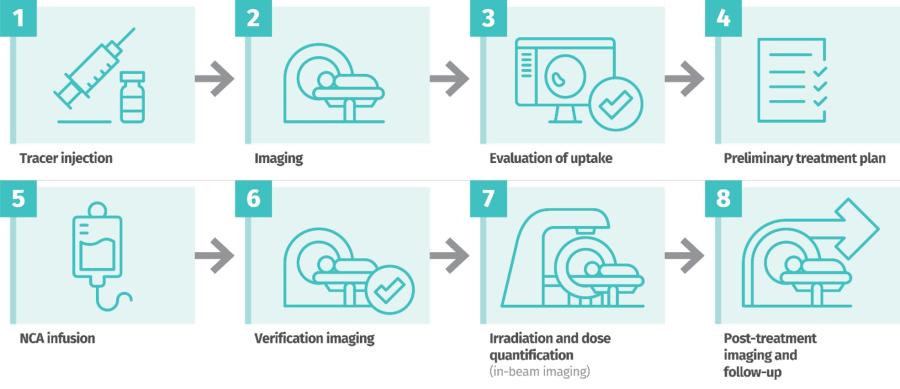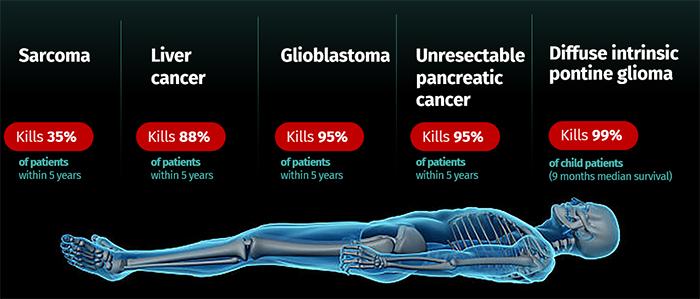
Neutron capture enhanced particle therapy (NCEPT) is a groundbreaking targeted radiotherapy technique pioneered by a multi-disciplinary team at ANSTO and its partner organisations. This innovative approach aims to treat cancers with poor prognosis, including brain cancer, pancreatic cancer and secondary melanoma. NCEPT enhances the effectiveness of particle therapy by combining the primary radiation beam (a proton or carbon ion beam) with a tumour-targeting neutron capture agent, which selectively boosts the radiation dose to cancer cells while minimising damage to healthy tissues.
ANSTO’s sovereign capabilities, including accelerators, neutron and photon sources and imaging technology along with expertise in imaging, particle physics, radiobiology, radiochemistry and therapeutics are crucial to the development and advancement of NCEPT.
By increasing the effectiveness of particle therapy, NCEPT has the potential to shortens treatment times, enabling healthcare providers to treat more patients and optimise the use of limited treatment facilities and resources. This could lead to lower costs per patient for the public health system.
NCEPT offers direct benefits to patients, especially children, by providing more effective radiotherapy through targeted radiation enhancement). This approach aims to reduce debilitating side-effects and late term complications associated with conventional radiotherapy.