
In Brief
-
The development of an effective drug to treat chronic lymphocytic leukaemia earned the Prime Minister's Prize for Science Innovation 2019
-
Investigators used macromolecular crystallography at the Australian Synchrotron to determine the 3D structure of a key protein that plays a role in minimising side effects of the drug
Professors Peter Czabotar, David Huang, Guillaume Lessene and Andrew Roberts have been recognised for their roles in the invention and development of venetoclax, an effective breakthrough anti-cancer drug with the Prime Minister's Prize for Innovation 2019.
This breakthrough research program led by the Walter and Eliza Hall Institute with international collaborators included investigations led by Peter Czabotar using the Macromolecular crystallography beamlines at the Australian Synchrotron.
The drug which triggers a signal in cancer cells causing them to die has been shown to be effective in treating chronic lymphocytic Leukaemia patients successfully.
The latest research paper about the molecular mechanism operating in the drug was published in Nature Communications in June this year.
"We were absolutely thrilled to hear the announcement of the prize to our colleagues, who have worked for many years to improve outcomes for CLL patients. Because our macromolecular beamlines are very useful in determining the structure of key proteins in cellular processes, they were important in this research," said Prof Andrew Peele, Director, Australian Synchrotron.
Read the media release from the Walter and Eliza Hall Institute
Original report on the research published in October 2014
The Australian Synchrotron has played a crucial role in the discovery of a new cancer drug now in advanced clinical trials in Melbourne for the treatment of chronic lymphocytic leukaemia (CLL).
Each year in Australia, around 350 people die from CLL and 1300 new cases are diagnosed, making this the nation’s most common leukaemia. Eighty per cent of CLL patients are aged 60 or older.
The research was performed at the Walter and Eliza Hall Institute (WEHI) in partnership with two major international pharmaceutical companies. WEHI structural biologist Dr Peter Czabotar said the Synchrotron was “key to discovering the drug”.

Dr Tom Caradoc-Davies (left), iPrincipal Scientist of the Macromolecularcrystallography beamlines with Dr Peter Czabotar, from the Walter and Eliza Hall Institute of Medical Research (WEHI),
“The highly detailed 3D images of protein structures that the Synchrotron provides were crucial,” Peter said.
“We needed this level of detail to develop a drug that targets one single protein – and not the other four very similar proteins in the same family – critical for minimising potential side-effects.”
The drug discovery and development work have taken five to 10 years to reach the trial stage. It was underpinned by more than 20 years of basic biology research, dating back to WEHI’s discovery that the BCL-2 family of proteins protects cells against apoptosis – the ‘programmed cell death’ process that the body uses to remove old or dangerous cells.
“It’s taken a long time to fully understand the function of BCL-2 proteins and turn this basic knowledge into drugs that can stop cancer cells from progressing into cancer,” Peter said.
“If we started a similar project now, our access to the Synchrotron would rapidly accelerate the identification of suitable drug targets and the development of potential drug candidates for clinical trials.
“Australian Synchrotron staff keep their beamlines in top shape, and their scientific skills help us solve protein structures and develop compounds to stop target proteins in their tracks.”
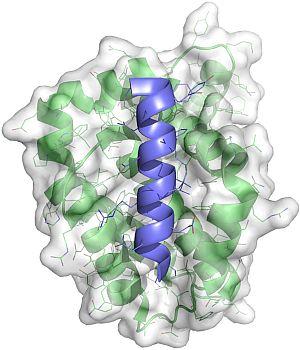
Structure of a Bcl-2 family protein bound to a natural BH3 ligand
Head of Business Development at WEHI, Julian Clark added: “the Synchrotron was instrumental in enabling researchers to achieve world-class synchrotron technical skills and attract major industry partners, contributing to Australia’s growing ability to host clinical trials for important new drugs”.
“This drug is potentially a major advance for patients with CLL that doesn’t respond adequately to currently available treatments,” according to Prof. Andrew Roberts, WEHI Head of Translational Research. “Early-stage clinical trials suggest that the drug may lead to complete remission in one-quarter of patients with advanced CLL that isn’t responding to other therapies. The clinical trials now underway will take at least three years.”
If the advanced clinical trials are successful, the CLL drug will be developed for market, to benefit cancer patients globally. The CLL work may also assist development of treatments for breast cancer and other diseases where cell death is not occurring at the appropriate rate.


