
ANSTO researchers have used nuclear techniques to assist in determining the mechanism behind the effectiveness of a potential new treatment for the childhood cancer neuroblastoma.

|
| Radiochemist Giancarlo Pascali and Imaging Physiologist Arvind Parmar used 64Cu with PET- CT to investigate the effectiveness of a new childhood cancer drug in a collaboration with Children's Cancer Institute |
Giancarlo Pascali and his ANSTO colleagues assisted researchers from the Children’s Cancer Institute(CCI) in Sydney, confirming that the anti-cancer effects of Dextran-Catechin (DC) on neuroblastoma cells are caused by modulation of the intracellular copper level.
The pilot study involved administering DC or saline (i.e. control) into the animal model of neuroblastoma to see if there was any statistical difference in intracellular copper levels in the tumours between control and treated groups.“PET-CT imaging using 64Cu-copper provided evidence that the drug under evaluation modulates the levels of copper in vivo in animal models of neuroblastoma tumour cells,” said Pascali, who is an international expert in radiochemistry and microfluidics.
Neuroblastoma is an aggressive cancer that accounts for approximately 40 per cent of infant deaths in Australia, and it is generally diagnosed only when already in metastatic phase. At that point, the only treatment, which is not always successful, is aggressive chemotherapy, which causes serious long term side effects in the children.
Previous research by Orazio Vittorio, Maria Kavallaris et al. using in vitro and in vivo techniques had confirmed that the new bioconjugate DC was effective in killing neuroblastoma cancer cells without harming normal cells, by modifying the copper level in tumour cells.
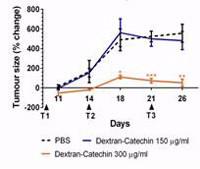
|
| Reduction of tumour growth in a neuroblastoma xenograft with Dextran catechin treatment (Vittorio O et al) |
Several other experimental proofs led to the suspicion that copper regulation (i.e. decrease) was implicated in the therapeutic mechanism of DC. This drug is a compound made up of Catechin, a natural antioxidant present in green tea, conjugated with Dextran; this formulation allows the anti-cancer activity of Catechin to be expressed, while protecting it from in vivo degradation.
In order for DC to make it into trials in humans, it is necessary to have a fully elucidated explanation of its therapeutic mechanism and evidence of its safety; therefore, studies improving this knowledge will speed up the utilization of this drug in actual treatments.
“We came up with the idea of using simple 64Cu-copper chloride (64CuCl2) in vivo to verify the reduction of intracellular copper by the drug treatment hypothesis” said Pascali.
The preliminary findings, which were reported by Pascali at the International Nanomedicine Conference held in Sydney in late June this year, confirmed an increased accumulation of copper in the tumour cells that was significantly reduced in the subjects treated with DC.
These findings were presented at the EANM conference in Barcelona in October 2016 and more recent results, from a larger campaign of experiments that confirmed the previous findings, will be presented in Melbourne at the international Theranostics World Congress in November.
Pascali said, “The64Cu-CuCl2 imaging approach was useful in understanding the curative mechanism of DC and could also be employed to determine whether DC can be used in other types of tumours, such as prostate cancer and glioblastoma, where initial animal testing of DC has been favourable.”
This project was championed by Giancarlo Pascali and Imaging Physiologist Arvind Parmar (above right), who carried out the experiments and PET CT imaging.
The animal model, the compound and the neuroblastoma cells were provided by Orazio Vittorio and Maria Kavallaris, scientists from the CCI. The radiotracer for these experiments was prepared by Radiometals Specialist Nigel Lengkeek.
“Using new reaction techniques and complex radiolabelling approaches, such as microfluidics, is something we excel at in Australia and has allowed our group to gain international recognition and visibility.
In this case, we were able to obtain deep understanding of the drug mechanism by employing a simple and underestimated radiotracer with a different aim in mind,” said Pascali.
