Published by NDF staff 22nd November, 2022
The favourable properties of ionic liquids make them useful for numerous applications.
High stability means ionic liquids are suitable for use as electrolytes in extreme environments such as space. The NDF have produced several selectively deuterated ionic liquids that have been used to increase our understanding of ionic liquids.
As powerful solvents, deuterated ionic liquids have allowed studies into the dissolution of cellulose, where insolubility has traditionally made processing difficult.
- Ionic liquids have numerous applications as powerful solvents and electrolytes. As such, they are candidates for renewable energy storage devices such as lithium-ion batteries.
- Alkyl methylimidazolium-based ionic liquids exhibit several useful properties, including high thermal stability and very low vapour pressure. They have been used as electrolytes in lithium-ion batteries and are useful lubricants.
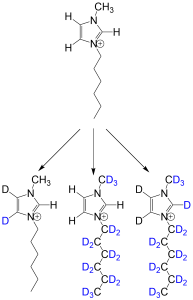
- Alkyl methylimidazolium-based ionic liquids can be reliably deuterated under hydrothermal conditions. Based on the reaction conditions used, the level of deuteration can be carefully controlled.
- To increase understanding of ionic liquids as highly stable electrolytes, the fine structural properties of the electrical double layer formed at the electrode-ionic liquid interface have been studied. The NDF has assisted international researchers (from CROSS, Japan Atomic Energy Agency and High Energy Accelerator Research Organization) by producing a number of selectively deuterated ionic liquids to achieve sufficient contrast for neutron reflectometry experiments. Data were obtained on the ordering, orientation and thickness of the charged layer structure.

The chemical structures of deuterated isotopologues of hexyl-methylimidazolium species synthesised by NDF.
These ionic liquids were used in neutron reflectometry experiments.1
- Another area of research with these ionic liquids is the dissolution of cellulose. Cellulose is an abundant and renewable polymer which is traditionally used in textiles and paper and has further technological applications in biomedicine and nanomaterials. However, its insolubility makes processing difficult. These types of ionic liquids can dissolve cellulose efficiently and safely.
- The NDF synthesised and provided deuterated alkyl methylimidazolium-based ionic liquids (here, ethyl chained, and acetate anion) to researchers (from BioPRIA - Monash and Technion-Israel Institute of Technology) for small angle neutron scattering experiments to aid with contrast variation.
- It is now understood that the ionic liquid penetrates the cellulose fibrils and strong adsorption disrupts interchain hydrogen bonding. This effectively makes a charged surface around the chains which prevents them from coming back together.
- These methylimidazolium-based ionic liquids, due to their powerful ability to dissolve cellulose, enable solution studies of cellulose. Deuteration of the ionic liquids helps with versatile neutron scattering experiments.
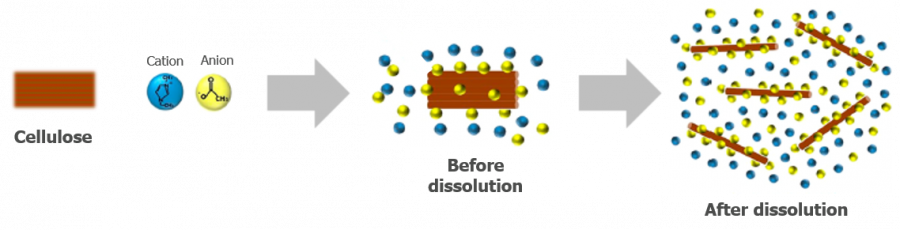
Ionic liquids effectively dissolve cellulose polymer chains, with a charged surface around the chains preventing them coming back together.3
NDF staff contributing to this work: Marina Cagnes, Dr Rob Russell
References
-
Controlled deuterium labelling of imidazolium ionic liquids to probe the fine structure of the electrical double layer using neutron reflectometry.
Akutsu-Suyama, K., Cagnes, M., Tamura, K., Kanaya, T. & Darwish, T. A.
Physical Chemistry Chemical Physics, 2019, 21, 17512-17516. -
Penetration behavior of an ionic liquid in thin-layer silica coating: Ionic liquid deuteration and neutron reflectivity analysis.
Akutsu, K., Cagnes, M., Niizeki, T., Hasegawa, Y. & Darwish, T. A.
Physica B: Condensed Matter, 2018, 551, 262-265. -
Cellulose Dissolution in Ionic Liquid: Ion Binding Revealed by Neutron Scattering.
Raghuwanshi, V. S., Cohen, Y., Garnier, G., Garvey, C. J., Russell, R. A., Darwish, T. & Garnier, G.
Macromolecules, 2018, 51, 7649-7655.


