A collaboration of ANSTO scientists led by the National Deuteration Facility (NDF) has developed a new screening method to rapidly determine if a deuterated molecule has improved metabolic stability. The research was published in Analytica Chimica Acta and is part of a post-doctoral research project of Dr Rhys Murphy, a lead author on the paper.
Because molecules labelled with deuterium (a non-radioactive isotope of hydrogen) can be metabolised more slowly than their non-deuterated (protonated) counterparts, they have a range of potential benefits.
In addition to the possibility of lowering drug doses, deuterated molecules can alter the type and quantity of metabolites that are formed.
The focus on these so-called “heavy” drugs has been renewed since the world’s first deuterated drug was approved by the U.S. Food and Drug Administration in 2017.
These benefits apply to radiotracers, which are molecules specifically designed for medical imaging to diagnose and track disease.
If the site of radiolabel attachment in the tracer (which acts as a beacon in the imaging process) is metabolically attacked too quickly, the image may not be informative.
In the study, the radiotracer, PBR111, which was developed at ANSTO for imaging neuroinflammation, was deuterated on the fluoropropoxy chain.
"It is not always clear where to deuterate in a molecule to gain an advantage. The analysis can be complex. Because the physical chemistry of both compounds is almost identical, separate analyses for both analogues are typically needed,” said Dr Tamim Darwish, a senior author and Manager of Operations at NDF.
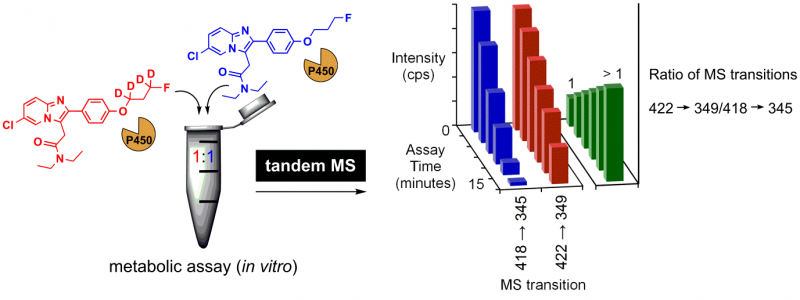
The team validated a one-pot method, in which the deuterated and non-deuterated analogues were mixed together and subjected to in vitro metabolism. The mixture was analysed by tandem mass spectrometry (MS) at different time intervals.
Caption: (Left) One-pot metabolic assay of a 1:1 mixture of deuterated and non-deuterated molecule. (Right) monitoring signals by tandem MS for both analogues (blue and red bars). A deviation of the ratio (green bars) from the initial value with assay time indicates a difference between the metabolism of the deuterated and non-deuterated molecules.
Using the ratio between corresponding MS signals, the improved metabolic stability was attributed to deuteration on the fluoropropoxy chain.
“As the distinct mass (unique to each molecule) was monitored, there was no need to analyse the compounds independently. Since the two compounds are part of the same mixture, there is consistency during the comparison,” said Darwish.
internal collaboration also involved the expertise of human health and biosciences researchers Dr Andrew Wotherspoon (co-lead author), Dr Benjamin Fraser, Dr Naomi Wyatt, Dr Nageshwar Yepuri (NDF) and
Prof Peter Holden (NDF Director).
“The method quickly tells you if the location of deuteration is having a positive impact on metabolism and if further studies may be warranted, perhaps in vivo” said Murphy, who presented the work at an international isotopes conference in Prague last year.
The method also confirms if deuteration has had no effect on metabolism before further time and resources are invested.
The authors report that their versatile approach could be applied as a rapid screening tool for analogues of other molecules, including pharmacologically-active molecules, to determine their suitability for deuteration.
“The new method can also be used with “cold” radiotracers, the stable isotope labelled analogue of a radiotracer, which makes handling easier,” said Fraser.
The NDF, a world-leading facility for chemical and biological deuteration, is partly supported by the National Collaborative Research Infrastructure Strategy – an initiative of the Australian Government.
This research was funded by a Research Attraction and Acceleration Program grant from the New South Wales Government.
DOI: https://www.sciencedirect.com/science/article/pii/S0003267019302016
