ANSTO supplies a wide range of nuclear medicines via our licensed facilities.
All our nuclear medicine products are manufactured at ANSTO in Good Manufacturing Practice (GMP) licensed clean rooms. Our facilities are licensed by the Therapeutic Goods Administration. Our adherence to strict quality assurance and control ensures the safety and efficacy of the products.
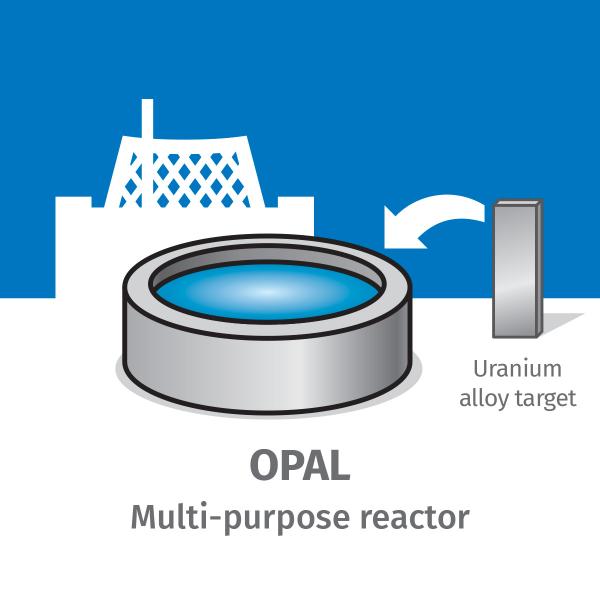
Midway through 2019, ANSTO began manufacturing molybdenum-99 (Mo-99) through a new state-of-the-art nuclear medicine manufacturing facility.
ANSTO's Mo-99, which decays to form Tc-99m, is produced at our Lucas Heights precinct and delivered to medical practices all over Australia. We are one of only a few organisations globally capable of delivering Tc-99m from one site.